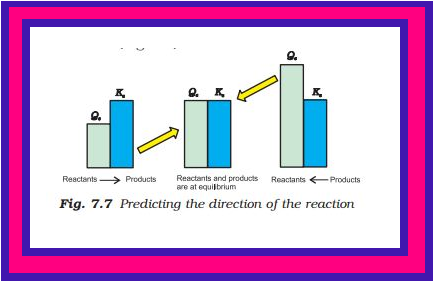
Predicting the Extent of a Reaction
`=>` The numerical value of the equilibrium constant for a reaction indicates the extent of the reaction.
`=>` But it is important to note that an equilibrium constant does not give any information about the rate at which the equilibrium is reached.
`=>` The magnitude of `color{red}(K_c)` or `color{red}(K_p)` is directly proportional to the concentrations of products (as these appear in the numerator of equilibrium constant expression) and inversely proportional to the concentrations of the reactants (these appear in the denominator). This implies that a high value of `color{red}(K)` is suggestive of a high concentration of products and vice-versa.
We can make the following generalisations concerning the composition of equilibrium mixtures:
• If `color{red}(K_c > 10^3)`, products predominate over reactants, i.e., if `color{red}(K_c)` is very large, the reaction proceeds nearly to completion. Consider
the following examples:
(a) The reaction of `color{red}(H_2)` with `color{red}(O_2)` at `500 K` has a very large equilibrium constant , `color{red}(K_c = 2.4 × 10^(47))`.
(b) `color{red}(H_2(g) + Cl_2(g) ⇌ 2HCl(g))` at 300K has `color{red}(K_c = 4.0 × 10^(31))`.
(c) `color{red}(H_2(g) + Br_2(g) ⇌ 2HBr (g))` at 300 K, `color{red}(K_c = 5.4 × 10^(18))`
• If `color{red}(K_c < 10^(–3)),` reactants predominate over products, i.e., if `color{red}(K_c)` is very small, the reaction proceeds rarely. Consider the following examples:
(a) The decomposition of `color{red}(H_2O)` into `color{red}(H_2)` and `color{red}(O_2)` at 500 K has a very small equilibrium constant, `color{red}(K_c = 4.1 × 10^(–48))`
(b) `color{red}(N_2(g) + O_2(g) ⇌ 2NO(g)),` at `298 K` has `color{red}(K_c = 4.8 ×10^(–31))`.
• If `color{red}(K_c)` is in the range of `color{red}(10^(–3))` to `color{red}(10^3),` appreciable concentrations of both reactants and products are present. Consider the following examples:
(a) For reaction of `color{red}(H_2)` with `color{red}(I_2)` to give `color{red}(HI, K_c = 57.0)` at `700K.`
(b) Also, gas phase decomposition of `color{red}(N_2O_4)` to `color{red}(NO_2)` is another reaction with a value of `color{red}(K_c = 4.64 × 10^(–3))` at `25°C` which is neither too small nor too large. Hence, equilibrium mixtures contain appreciable concentrations of both `color{red}(N_2O_4)` and `color{red}(NO_2).`
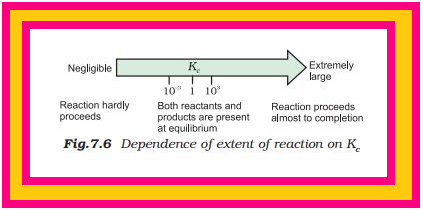
These generarlisations are illustrated in Fig. 7.6
`=>` But it is important to note that an equilibrium constant does not give any information about the rate at which the equilibrium is reached.
`=>` The magnitude of `color{red}(K_c)` or `color{red}(K_p)` is directly proportional to the concentrations of products (as these appear in the numerator of equilibrium constant expression) and inversely proportional to the concentrations of the reactants (these appear in the denominator). This implies that a high value of `color{red}(K)` is suggestive of a high concentration of products and vice-versa.
We can make the following generalisations concerning the composition of equilibrium mixtures:
• If `color{red}(K_c > 10^3)`, products predominate over reactants, i.e., if `color{red}(K_c)` is very large, the reaction proceeds nearly to completion. Consider
the following examples:
(a) The reaction of `color{red}(H_2)` with `color{red}(O_2)` at `500 K` has a very large equilibrium constant , `color{red}(K_c = 2.4 × 10^(47))`.
(b) `color{red}(H_2(g) + Cl_2(g) ⇌ 2HCl(g))` at 300K has `color{red}(K_c = 4.0 × 10^(31))`.
(c) `color{red}(H_2(g) + Br_2(g) ⇌ 2HBr (g))` at 300 K, `color{red}(K_c = 5.4 × 10^(18))`
• If `color{red}(K_c < 10^(–3)),` reactants predominate over products, i.e., if `color{red}(K_c)` is very small, the reaction proceeds rarely. Consider the following examples:
(a) The decomposition of `color{red}(H_2O)` into `color{red}(H_2)` and `color{red}(O_2)` at 500 K has a very small equilibrium constant, `color{red}(K_c = 4.1 × 10^(–48))`
(b) `color{red}(N_2(g) + O_2(g) ⇌ 2NO(g)),` at `298 K` has `color{red}(K_c = 4.8 ×10^(–31))`.
• If `color{red}(K_c)` is in the range of `color{red}(10^(–3))` to `color{red}(10^3),` appreciable concentrations of both reactants and products are present. Consider the following examples:
(a) For reaction of `color{red}(H_2)` with `color{red}(I_2)` to give `color{red}(HI, K_c = 57.0)` at `700K.`
(b) Also, gas phase decomposition of `color{red}(N_2O_4)` to `color{red}(NO_2)` is another reaction with a value of `color{red}(K_c = 4.64 × 10^(–3))` at `25°C` which is neither too small nor too large. Hence, equilibrium mixtures contain appreciable concentrations of both `color{red}(N_2O_4)` and `color{red}(NO_2).`
These generarlisations are illustrated in Fig. 7.6